JAMB Chemistry 2018
A given amount of gas occupies 10.0dm3 at 4atm and 273°C. The number of moles of the gas present is [Molar volume of gas at s.t.p = 22.4dm3].
Correct Answer: A. 0.89 mol
According to Charles’ law, the volume of a gas becomes zero at
Correct Answer: C. -273°C
A substance that is used as a ripening agent for fruits is
Correct Answer: A. Ethene
The Sulphide which is insoluble in dilute hydrochloric acid is
Correct Answer: B. CuS
What is the pH of 0.001 moldm-3 solution of sodium hydroxide?
Correct Answer: D. 11
The type of bonding in [Cu(NH3)4]2+ is
Correct Answer: A. coordinate
Which of the following is an example of a chemical change?
Correct Answer: B. Rusting of iron
To what temperature must a gas at 273K be heated in order to double both its volume and pressure?
Correct Answer: D. 1092K
According to the Kinetic Theory, an increase in temperature causes the kinetic energy of particles to
Correct Answer: B. Increase
An element used in the production of matches is
Correct Answer: D. Sulphur
Which of the following gases may not be dried with Concentrated sulphuric acid?
Correct Answer: B. NH3
A correct electrochemical series can be obtained from Na, Ca, Al, Mg, Zn, Fe, Pb, H, Cu, Hg, Ag, Au by interchanging
Correct Answer: A. Al and Mg
A basic postulate of the kinetic theory of gases is that the molecules of a gas move in straight lines between collisions. This implies that
Correct Answer: B. Forces of repulsion exist
On which of the following is the solubility of a gaseous substance dependent? I. Nature of solvent II. Nature of solute III. Temperature IV. Pressure
Correct Answer: D. I, III and IV
Which of the following statements is correct about the periodic table?
Correct Answer: B. The valence electrons of the elements in the same period increase progressively across the period
The periodic classification is an arrangement of the elements in order of their increasing
Correct Answer: D. Atomic numbers
If 1 litre of 2.2M sulphuric acid is poured into a bucket containing 10 litres of water and the resulting solution mixed thoroughly, the resulting sulphuric acid concentration will be
Correct Answer: C. 0.22M
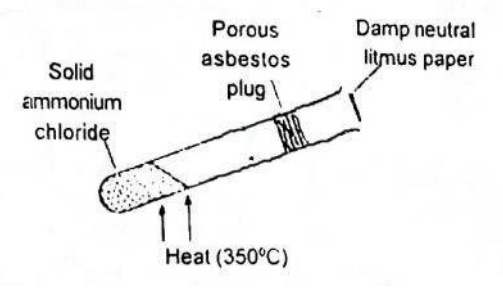
In the shown experiment above, the litmus paper will initially
Correct Answer: D. Turn blue
The boiling of fat and aqueous caustic soda is referred to as
Correct Answer: D. Saponification
Which of the following pairs of substances will react further with oxygen to form a higher oxide?
Correct Answer: D. SO2 and NO
In the preparation of oxygen by heating KClO3 in the presence of MnO2 only moderate heat is needed because the catalyst acts by
Correct Answer: C. Increasing the rate of the reaction
Methanoic acid mixes with water in all proportions and has about the same boiling point as water. Which of the following methods would you adopt to obtain pure water from a mixture of Sand, water and methanoic acid?
Correct Answer: A. Fractional distillation
A quantity of electricity liberates 3.6g of Silver from its salt. What mass of aluminium will be liberated from its salt by the same quantity of electricity? [Al = 27, Ag = 108].
Correct Answer: D. 0.3g
Suitable reagents for the laboratory preparation of nitrogen are
Correct Answer: A. Sodium dioxonitrate(III) and ammonium chloride
The number of electrons in the valence shell of an element of atomic number 14 is?
Correct Answer: D. 4
What volume of oxygen will remain after reacting 8cm3 of hydrogen gas with 20cm3 of oxygen gas?
Correct Answer: D. 16cm3
If one of the following oxides is heated with hydrogen or carbon using a bunsen burner, it is not reduced to the metal. Which one is it?
Correct Answer: B. Magnesium oxide
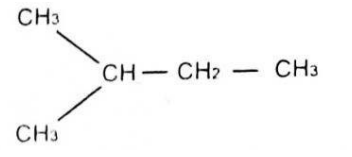
The IUPAC name for CH3-CH(CH3)-CH2-CH3 is
Correct Answer: C. 2-methylbutane
An aqueous solution of a metal salt, M, gives a white precipitate with NaOH which dissolves in excess NaOH. With aqueous ammonia, the solution of M also gives a white precipitate which dissolves in excess ammonia. Therefore the cation in M is
Correct Answer: A. Zn2+
What is the concentration of a solution containing 2g of NaOH in 100cm3 of solution? [Na = 23, O =16, H = 1]
Correct Answer: B. 0.50 moldm-3
How many atoms are present in 6.0g of magnesium? [Mg = 24, NA = 6.02 x 1023]
Correct Answer: D. 1.51 x 1022
The radioisotope used in industrial radiography for the rapid checking of faults in welds and castings is?
Correct Answer: C. Cobalt-60
Beryllium and Aluminum have similar properties because they
Correct Answer: D. Are positioned diagonally to each other
For the reaction mE + nF ⇌ pG + qH, the expression for the equilibrium constant is
Correct Answer: C. [G]p[H]q/[E]m[F]n
The reactions represented by the equations
below
demonstrate the
(I). 3CuO(s) + 2NH3(g) →
3Cu(s) +
3H2O(l) + N2(g)
(II).
2NH3(g) +
3Cl2(g) → 6HCl(g) + N2(g)
(III).
4NH3(g) + 3O2(g) → 6H2O(l)
+
N2(g)
Correct Answer: C. Reducing properties of ammonia
The salt that reacts with dilute hydrochloric acid to produce a pungent smelling gas which decolourizes acidified purple potassium tetraoxomanganate(VII) solution is
Correct Answer: B. Na2SO3
The refreshing and characteristic taste of soda water and other soft drinks is as a result of the presence of
Correct Answer: A. carbon(IV)oxide
Which of the following are mixtures?
I.
Petroleum
II. Rubber latex
III. Vulcanizer’s
solution
IV.
Carbon disulphide
Correct Answer: A. I, II and III
A balanced chemical equation obeys the law of
Correct Answer: A. Conservation of mass
When air which contains the gases Oxygen, nitrogen, carbondioxide, water vapour and the rare gases, is passed through alkaline pyrogallol and then over quicklime, the only gases left are;
Correct Answer: D. Nitrogen and the rare gases
In the laboratory preparation of oxygen, the gas cannot be collected by displacement of air because
Correct Answer: B. The density of air is nearly the same as that of oxygen
2KClO3(s) MnO2→
2KCl(s) +
3O2(g)
The importance of the catalyst in the
reaction
above is that
Correct Answer: D. The reaction takes place more rapidly at a lower temperature
Which of the following is used to power steam engines?
Correct Answer: B. Coal
In the reaction between sodium hydroxide and sulphuric acid solutions, what volume of 0.5 molar sodium hydroxide would exactly neutralise 10cm3 of 1.25 molar sulphuric acid?
Correct Answer: D. 50 cm3
To what volume must 300cm3 of 0.60M sodium hydroxide solution be diluted to give a 0.40M solution?
Correct Answer: A. 450 cm3
Diamond is a bad conductor of electricity because its bonding electrons are used in
Correct Answer: B. Covalent bond formation
The reaction between an organic acid and an alcohol in the presence of an acid catalyst is known as;
Correct Answer: C. Esterification
The constituent common to duralumin and alnico is
Correct Answer: C. Al
The solubility of a solid that dissolves in a given solvent with the liberation of heat will
Correct Answer: B. Decrease with an increase in temperature
What mass of Cu would be produced by the cathodic reduction of Cu2+ when 1.60A of current passes through a solution of CuSO4 for 1 hour. (F=96500Cmol-1, Cu=64)
Correct Answer: C. 1.91g